

Remote Temperature Monitoring Kit
TempTraq is an FDA Cleared Class II medical device that gives healthcare providers the first wearable, wireless, continuous body temperature monitor in the form of a soft, comfortable, disposable patch.
TempTraq is the only single use, US manufactured FDA cleared device for remote continuous temperature monitoring.
- Earlier Fever Detection
- Remote Patient Monitoring
- Real-time Continuous Temperature Monitoring (CTM)
- Pre-Configured Patient Kit with Instructions
- HIPAA Compliant Cloud with Electronic Health Record Integration
Earlier Detection of Fever May Lead to
- Improved patient outcomes
- Reduced hospitalizations
- Maximized revenues
- Improved patient satisfaction
- Improved MIPS quality score
- Reduced re-admissions
Population Risk Management—TempTraq helps healthcare organizations manage higher risk patients where earlier detection of fever provides significant benefits:
- Patients with high risk of infection
- Post operative
- Sepsis
- Oncology treatment
- CAR-T
- Immunotherapy with CRS
- COVID-19 and other infectious diseases

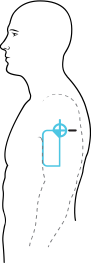
Axillary Temperature vs. Skin Temperature
Numerous studies including TempTraq clinical trials have demonstrated that axillary (armpit) temperature is a clinically accurate measurement of core body temperature. Devices placed elsewhere on the surface of the body measure skin temperature (not core) which can be effected by activity and surroundings.
Accuracy and Equivalency in Three Hospital Studies
• Cleveland Clinic
• University Hospitals Seidman Cancer Center
• Cincinnati Children’s Hospital
Earlier Fever Detection
In a clinical study conducted by University Hospitals Seidman Cancer Center, TempTraq was shown to detect temperature rise earlier than the Standard of Care by a median of 140.1 minutes (range of 30-180 minutes) with bone marrow transplant patients.
Technical Specifications
- Clearances: FDA 510k Cleared, CE, Health Canada, TGA
- Operating Life: 24 hours or 72 hours; Disposable/Single Use
- Accuracy: Conforms with ASTM E1112-00 (2011) +/-0.1°C or+/-0.2°F over the temperature range of the device
- Wireless: Bluetooth Receiving Device, Low Energy, patch must be within 40 feet of Bluetooth device
- Dimensions: 50 mm x 100 mm soft patch applied in the axillary position underarm
- Safe: For all ages and all skin types; does not contain materials made with natural latex rubber
- Temperature Display: Fahrenheit or Celsius
- Temperature Range: 87.0 °F -109.3 °F (30.6 °C -42.9 °C)
- Alerts: Audible and/or visual notifications of rising temperature via applications

Continuous
Once activated, TempTraq continuously monitors axillary temperature for up to 72 hours per patch, and issues alerts when temperature rises.
Accurate
Clinically tested and proven accuracy (in agreement with Pulmonary Artery Catheter).
Wireless
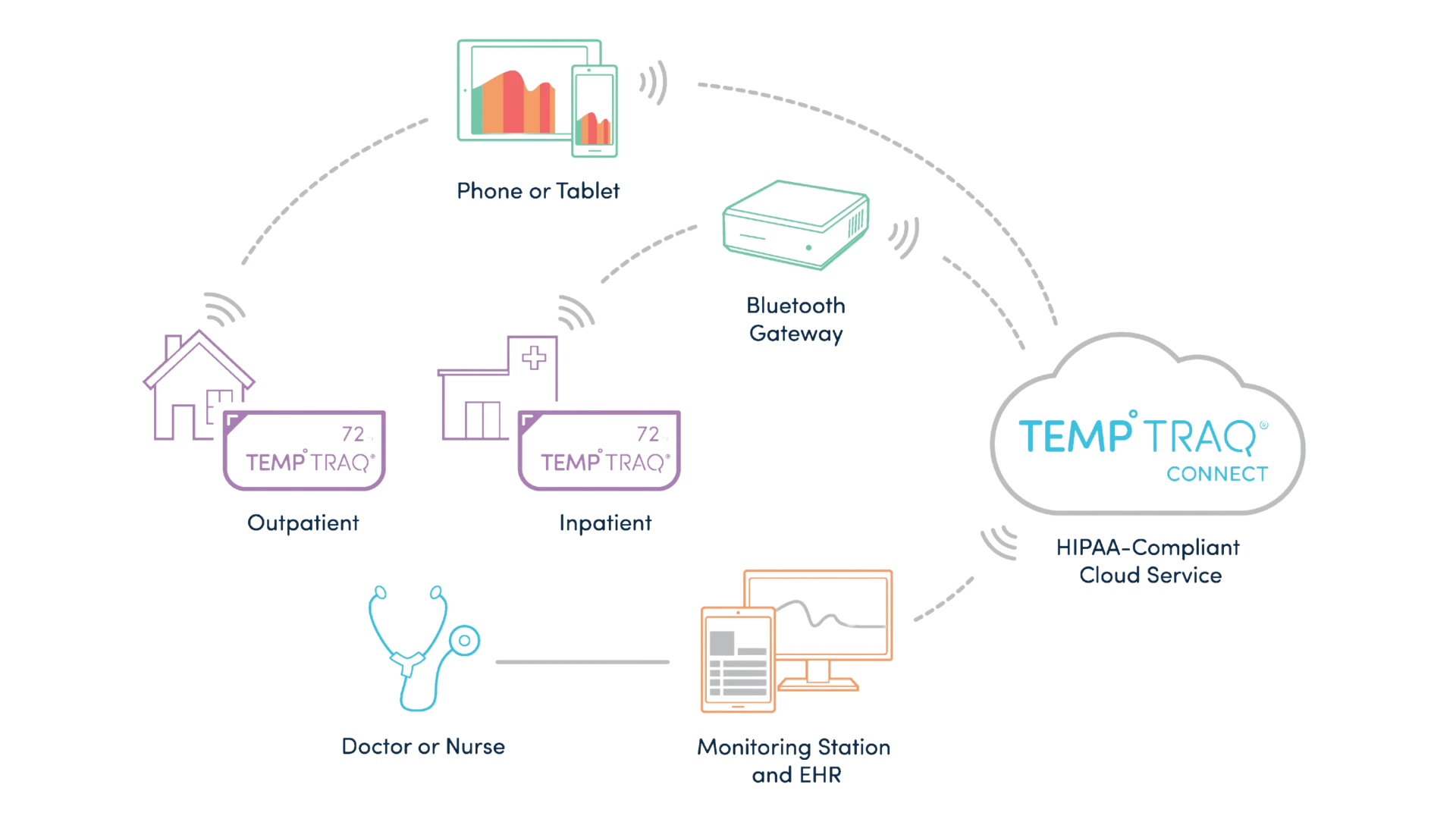
Data transmits via Bluetooth, allowing patient freedom of movement.
Non-Invasive
TempTraq applies easily to patient with a gentle adhesive.
Remote Monitoring
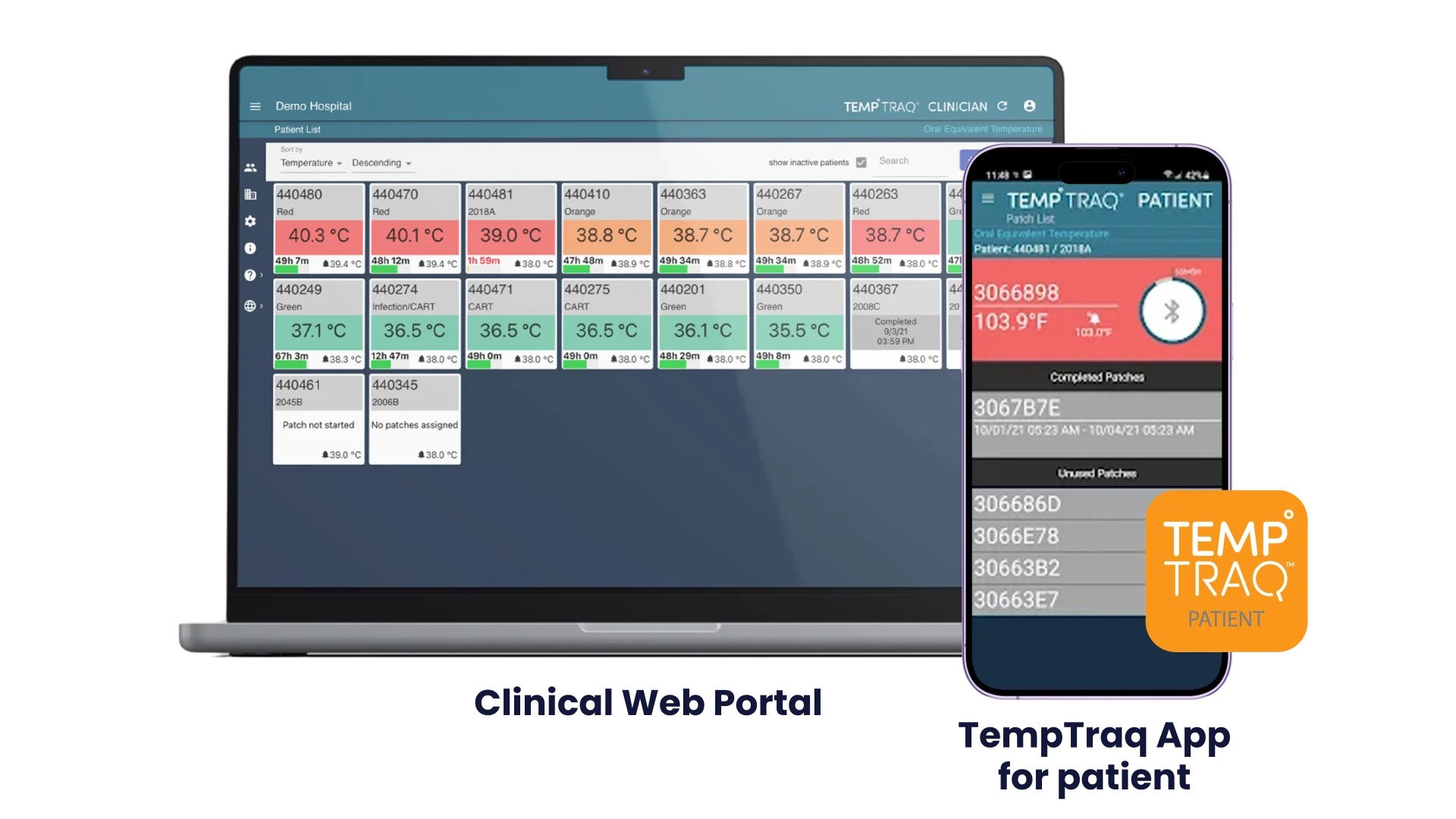
Patients can be remotely monitored via TempTraq Connect’s secure, HIPAA compliant cloud service.
Fully Integrated
TempTraq applies easily to patient with a gentle adhesive.


















